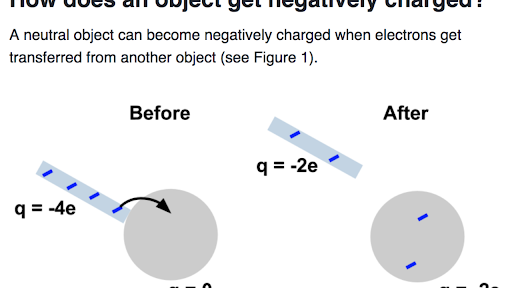
An Electrically Neutral Object Is Given a Negative Charge
Because the electron has such a small charge the coulomb abbreviated with a capital C is often used as unit of charge for 624 10 18 electrons. A small metal ball is given a negative charge then brought near ie within about 110 the length of the rod to end A of the rod Figure 1.
Conservation Of Charge Review Article Khan Academy
In an electrically neutral atom the number of protons in the nucleus is equal to the number of.
. There is now a. The electron is a subatomic particle denoted by the symbol e or β whose electric charge is negative one elementary charge. The chloride ion has a charge of -1.
The same number of electrons must be lost as gained so the resulting products will be electrically neutral after reaction. When ionic reactions take place the reactants are electrically neutral to begin with. Electrons that surround the nucleus.
A small object A electrically charged creates an electric field. Neutrons in the nucleus. Chlorine is neutral to begin with so if it gains an electron it will have a net charge of -1.
Assume that two of the electrons at the negative terminal have attached themselves to a nearby neutral atom. Electrons belong to the first generation of the lepton particle family and are generally thought to be elementary particles because they have no known components or substructure. Using Figure 3 explain in terms of Coulombs law why a polar molecule such as in Figure 3 is attracted by both positive and negative charges.
Given waters polar character explain what effect humidity has on removing excess charge from objects. A positive charge and a negative charge held near each other are released. Because electrons carry a negative charge and a coulomb refers to a positive charge some definitions are needed.
As they move the force on each particle. In these units 1 ampere A is a current created by 1 coulomb C passing per second. The electron has a mass that is approximately 11836 that of the.

Comments
Post a Comment